FoldersSynchronizer 5.1 macOS
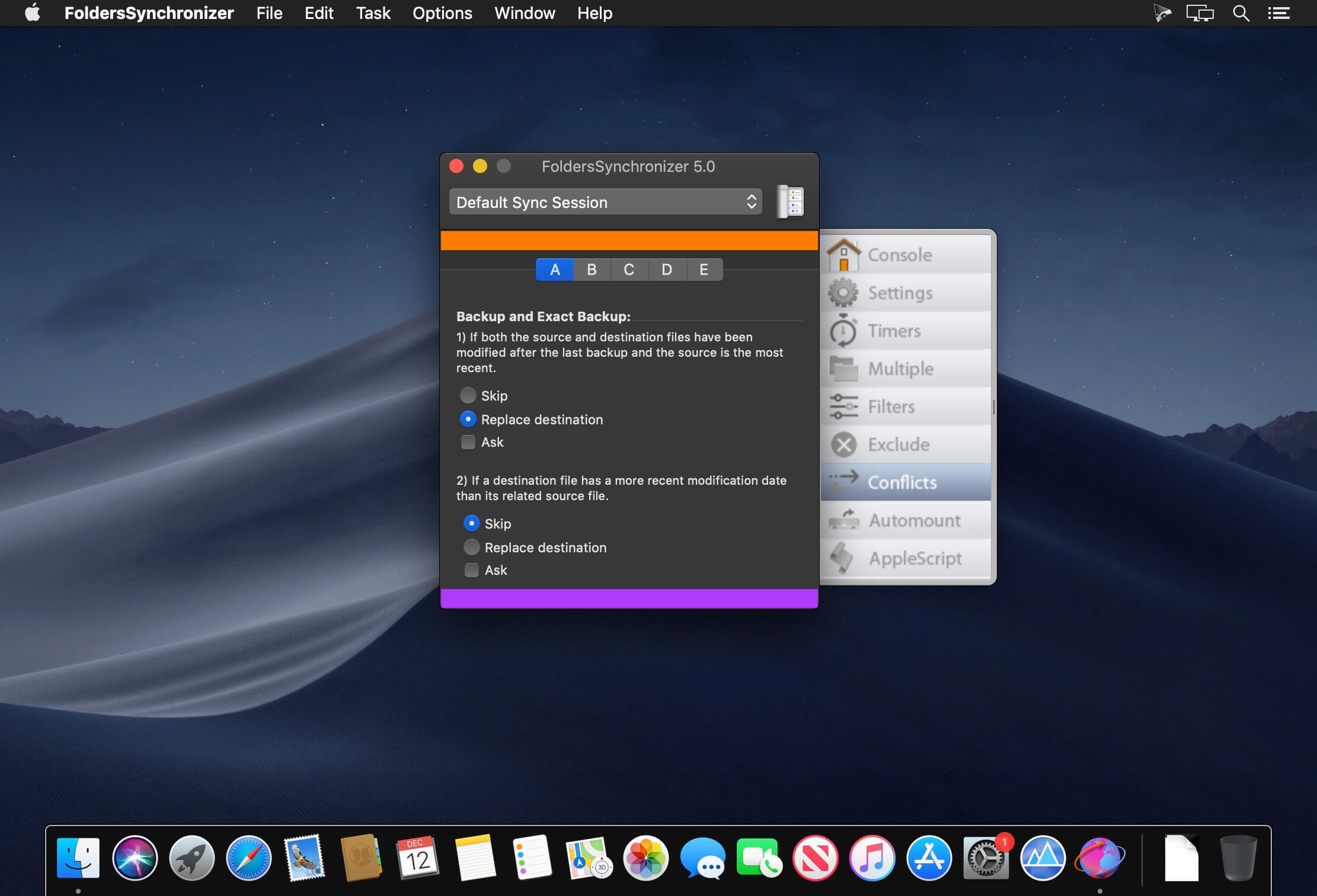
FoldersSynchronizer is a popular and useful utility that synchronizes and backs-up files, folders, disks and boot disks. On each session you can apply special options like Timers, Multiple Folders, Filters, Exclude Items, Auto-Mount local and remote volumes, launch your own AppleScripts, set how to resolve conflicts, execute an incremental or an exact copy, include locked files, and more.
What’s New:
Version 5.1.0:
- This is a 64 bit version, successfully tested on macOS 10.15 Catalina.
- Tuned up for retina displays.
- New icons, look and feel for both light and dark mode.
- Other minor fixes and improvement.
- Free upgrade for version 5 registered users.
Pounds to Grams How to convert Grams to Pounds. 1 gram (g) is equal to 0.2185 pounds (lbs). 1 g = 0.2185 lb. The mass m in pounds (lb) is equal to the mass m in grams (g) divided by 453.59237. FoldersSynchronizer is the best and most popular utility for Mac to Synchronize and Backup files, folders, disks and boot disks. Available on 64bits with Timers, Multiple Folders, Filters, Exclude, Conflicts, Auto-mount Volumes, AppleScript and Settings. FoldersSynchronizer can sync/backup the deleted files, run with the root permissions, create working sessions, save/open your settings files. Image Results for '5 White And Round' Below are results that match your criteria. Click on an image or generic/brand name to search for information about the drug in our database. Grams to mg How to convert Milligrams to Grams. 1 milligram (mg) is equal to 1/1000 grams (g). 1 mg = (1/1000) g = 0.001 g. The mass m in grams (g) is equal to the mass m in milligrams (mg) divided by 1000. GDevelop 5 is an open-source, cross-platform game engine designed for everyone - it's extensible, fast and easy to learn. To use GDevelop 5, you must activate JavaScript in your b.
Compatibility: macOS 10.13 or later 64-bit
Homepagehttp://www.softobe.com/
Homepagehttp://www.softobe.com/
Screenshots
Half-Life Problems #1 - 10
| Ten Examples | Problems involving carbon-14 | |
| Probs 11-25 | Problems involving uranium-238 | |
| Probs 26-40 | Examples and Problems only (no solutions) | Return to Radioactivity menu |
Problem #1: The half-life of Zn-71 is 2.4 minutes. If one had 100.0 g at the beginning, how many grams would be left after 7.2 minutes has elapsed?
Solution:
7.2 / 2.4 = 3 half-lives(1/2)3 = 0.125 (the amount remaining after 3 half-lives)
100.0 g x 0.125 = 12.5 g remaining
Problem #2: Pd-100 has a half-life of 3.6 days. If one had 6.02 x 1023 atoms at the start, how many atoms would be present after 20.0 days?
Solution:
20.0 / 3.6 = 5.56 half-lives(1/2)5.56 = 0.0213 (the decimal fraction remaining after 5.56 half-lives)
(6.02 x 1023) (0.0213) = 1.28 x 1022 atoms remain
Problem #3: Os-182 has a half-life of 21.5 hours. How many grams of a 10.0 gram sample would have decayed after exactly three half-lives?
Solution:
(1/2)3 = 0.125 (the amount remaining after 3 half-lives)Page booth 2 3 – website screenshot tool mac. 10.0 g x 0.125 = 1.25 g remain
10.0 g − 1.25 g = 8.75 g have decayed
Note that the length of the half-life played no role in this calculation. In addition, note that the question asked for the amount that decayed, not the amount that remaning.
Problem #4: After 24.0 days, 2.00 milligrams of an original 128.0 milligram sample remain. What is the half-life of the sample?
Solution:
The decimal fraction remaining:
2.00 mg / 128.0 mg = 0.015625
2) How many half-lives must have elaspsed to get to 0.015625 remaining?
(1/2)n = 0.015625n log 0.5 = log 0.015625
n = log 0.5 / log 0.015625
n = 6
3) Determine the half-life:
24 days / 6 half-lives = 4.00 days
Problem #5: A radioactive isotope decayed to 17/32 of its original mass after 60 minutes. Find the half-life of this radioisotope.
Solution:
17/32 = 0.53125 (this is the decimal amount that remains)(1/2)n = 0.53125
n log 0.5 = log 0.53125
n = 0.91254 (this is how many half-lives have elapsed)
60 min / 0.91254 = 65.75 min
n = 66 min (to two sig figs)
Problem #6: How long will it take for a 40.0 gram sample of I-131 (half-life = 8.040 days) to decay to 1/100 its original mass?
Solution:
(1/2)n = 0.01n log 0.5 = log 0.01
n = 6.64
6.64 x 8.040 days = 53.4 days
Problem #7: Fermium-253 has a half-life of 0.334 seconds. A radioactive sample is considered to be completely decayed after 10 half-lives. How much time will elapse for this sample to be considered gone?
Solution:
0.334 x 10 = 3.34 seconds
Problem #8: At time zero, there are 10.0 grams of W-187. If the half-life is 23.9 hours, how much will be present at the end of one day? Two days? Seven days?
Solution:
24.0 hr / 23.9 hr/half-life = 1.0042 half-livesOne day = one half-life; (1/2)1.0042 = 0.4985465 remaining = 4.98 g
Two days = two half-lives; (1/2)2.0084 = 0.2485486 remaining = 2.48 g
Seven days = 7 half-lives; (1/2)7.0294 = 0.0076549 remaining = 0.0765 g
Problem #9: 100.0 grams of an isotope with a half-life of 36.0 hours is present at time zero. How much time will have elapsed when 5.00 grams remains?
Window tidy 2 1 4 – manage windows with ease. Solution:
5.00 / 100.0 = 0.05 (decimal fraction remaining)(1/2)n = 0.05
n log 0.5 = log 0.05
n = 4.32 half-lives
36.0 hours x 4.32 = 155.6 hours
Problem #10: How much time will be required for a sample of H-3 to lose 75% of its radioactivity? The half-life of tritium is 12.26 years.
Hype pro 3 6 8 plus. Solution:
(1/2)n = 0.25
n = 2 (remember (1/2)2 = 1/4 and 1/4 = 0.25)
12.26 x 2 = 24.52 years
Comment: the more general explanation follows:
(1/2)n = 0.25n log 0.5 = log 0.25
n = log 0.25 / log 0.5
Folderssynchronizer 5 0 G Ml Represents
n = 2
Folderssynchronizer 5 0 Gold
| Ten Examples | Problems involving carbon-14 | |
| Probs 11-25 | Problems involving uranium-238 | |
| Probs 26-40 | Examples and Problems only (no solutions) | Return to Radioactivity menu |
